
Simulations revealed that protons diffuse mainly along the interfaces of LuO 6 layers, which form cubic close-packed c BaO 3 layers, rather than through the AlO 4 layers. The second important characteristic is related to how protons move through Ba 2LuAlO 5. In turn, the oxide's higher water content increases its proton conductivity through various mechanisms, such as higher proton concentration and enhanced proton hopping. This large water uptake, which occurs within two opposing layers of AlO 4 tetrahedra, is made possible by a high number of intrinsic oxygen vacancies in the hexagonal close-packed h' BaO layers. The first is that this oxide absorbs a large quantity of water (H 2O), compared to other similar materials, to form Ba 2LuAlO 5*xH 2O (with x = 0.50). Through molecular dynamics simulations and neutron diffraction measurements, they learned two important characteristics of Ba 2LuAlO 5. Later, the team sought to find out the underlying reasons for this property. Experiments on Ba 2LuAlO 5 samples revealed that this material has a high proton conductivity in its bulk at low temperatures-its conductivity was 10 ‒2 S cm ‒1 at 487☌ and 1.5×10 ‒3 S cm ‒1 at 232☌-even without additional chemical refinements, such as doping. This was motivated by the results of previous studies highlighting the importance of these vacancies in proton conduction. Yashima and colleagues discovered Ba 2LuAlO 5 while focusing on finding compounds with numerous intrinsic oxygen vacancies. In their latest study, published in Communications Materials, the team reported the remarkable properties of Ba 2LuAlO 5, a new hexagonal perovskite-related oxide that has provided interesting insights into proton conduction. To address this challenge, a team of researchers, including Professor Masatomo Yashima from Tokyo Institute of Technology (Tokyo Tech) in Japan, has been on the lookout for good proton conductor candidates for PCFCs. Unfortunately, only a few proton-conducting materials with reasonable performance are currently known, which is slowing down progress in this field. Thanks to a much lower operating temperature in the range of 300 to 600☌, PCFCs can ensure a stable energy supply at a lower cost, compared to most other fuel cells. These cells use special ceramics that conduct protons (H +) instead of oxide anions (O 2−). That is why many scientists have focused on protonic ceramic fuel cells (PCFCs) instead. However, typical fuel cells based on solid oxides have a notable drawback in that they operate at high temperatures, usually over 700☌. 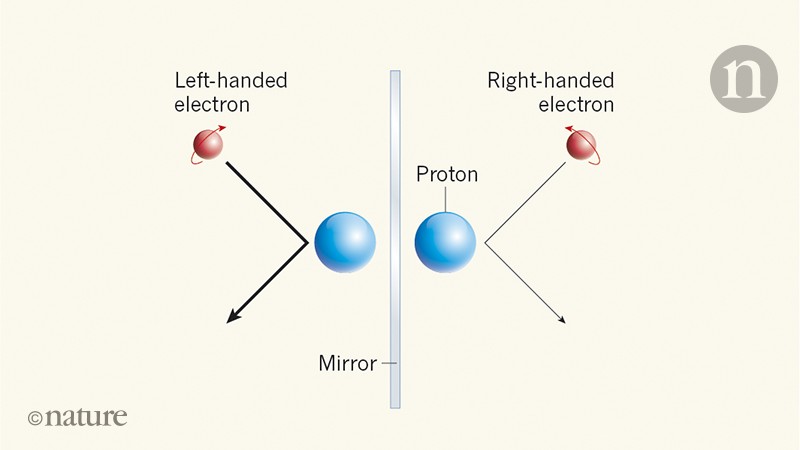 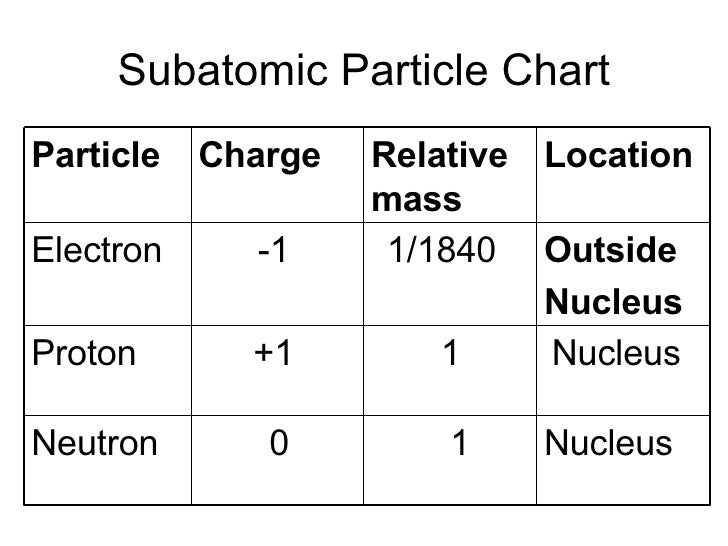
Among them, fuel cells have been steadily gaining traction since the 1960s as a promising approach to producing electricity directly from electrochemical reactions. Eager to eventually replace traditional energy sources such as coal and oil, scientists across the world are trying to develop environmentally friendly technologies that produce energy safely and more efficiently. 
Treating the charges on nitrogen and argon as the sums of charges on protons, electrons and neutrons, it is deduced that the proton charge is (1±4×10 −20) e and the charge on the neutron is less than 4 × −20 e.When talking about sustainability, the ways in which a society generates energy are some of the most important factors to consider. We have found that the charge on an argon atom (18 protons, 18 electrons and 22 neutrons) is not greater than 8×10 −20 e and that on a nitrogen molecule (14 protons, 14 electrons and 14 neutrons) is not greater than 12 × 10 −20 e. It therefore seemed desirable to find whether matter in which there is an excess of neutrons is electrically neutral. Since matter also contains neutrons, they have assumed in effect that the neutron has a charge equal to that of a hydrogen atom but the neutron might equally well have a charge opposite to that of a hydrogen atom, in which case their experiment does not settle the point at issue, since carbon dioxide contains equal numbers of protons and neutrons (to within 0.1 per cent). In fact, Piccard and Kessler 3 attempted to detect such a difference in 1925, and found that a molecule of carbon dioxide did not have an electric charge greater than 2×10 −19 e, where - e is the electronic charge, from which they concluded that the magnitudes of the proton and electron charges were the same to within 5 parts in 10 21, assuming that matter was built entirely of protons and electrons. IT has recently been suggested by Bondi and Ly ttleton 1,2 that the magnitudes of the electric charges on the proton and electron may differ by a little more than one part in 10 18, in which case electrostatic forces would cause the universe to expand. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
